
Lyme Disease Testing Market Report 2026
Global Outlook – By Test Type (Serological Tests, Nucleic Acid Tests, Urine Antigen Tests, Other Test Types), By Testing Methodology (Enzyme-Linked Immunosorbent Assay (ELISA), Weste Blot, Polymerase Chain Reaction (PCR), Immunofluorescence Assay (IFA), Other Emerging Technologies), By Disease Stage (Early Lyme Disease, Disseminated Lyme Disease, Chronic Lyme Disease, Asymptomatic Lyme Disease Detection), By Sample Type (Blood, Urine, Cerebrospinal Fluid(CSF), Other Blood Samples), By End-User (Hospitals And Clinics, Diagnostic Laboratories, Home Care Settings, Research Institutions, Public Health Organizations) – Market Size, Trends, Strategies, and Forecast to 2035
Lyme Disease Testing Market Overview
• Lyme Disease Testing market size has reached to $10.68 billion in 2025 • Expected to grow to $16.46 billion in 2030 at a compound annual growth rate (CAGR) of 9% • Growth Driver: Rising Prevalence Of Tick-Borne Illnesses Fueling The Need For Early Detection And Diagnosis • Market Trend: Technological Advancements In Diagnostic Tools For Enhanced Accuracy And Early Detection Capabilities • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Lyme Disease Testing Market?
Lyme disease testing refers to diagnostic procedures used to detect infection with Borrelia burgdorferi, the bacteria responsible for Lyme disease, typically transmitted through tick bites. The primary purpose of lyme disease testing is to accurately diagnose the infection, especially in individuals showing symptoms such as fever, fatigue, and the characteristic bull's-eye rash. The main test types involved in lyme disease testing include serological tests, nucleic acid tests, urine antigen tests, and others. Serological tests are diagnostic procedures that detect antibodies or antigens in a person’s blood to determine exposure to infections or immune responses. Various testing methodologies comprise enzyme-linked immunosorbent assay (ELISA), Western blot, polymerase chain reaction (PCR), immunofluorescence assay (IFA), and others. Different disease stages include early Lyme disease, disseminated Lyme disease, chronic Lyme disease, and asymptomatic Lyme disease detection. The different sample types used for testing include blood, urine, cerebrospinal fluid (CSF), and others. These tests are utilized by various end-users, such as hospitals and clinics, diagnostic laboratories, home care settings, research institutions, and public health organizations.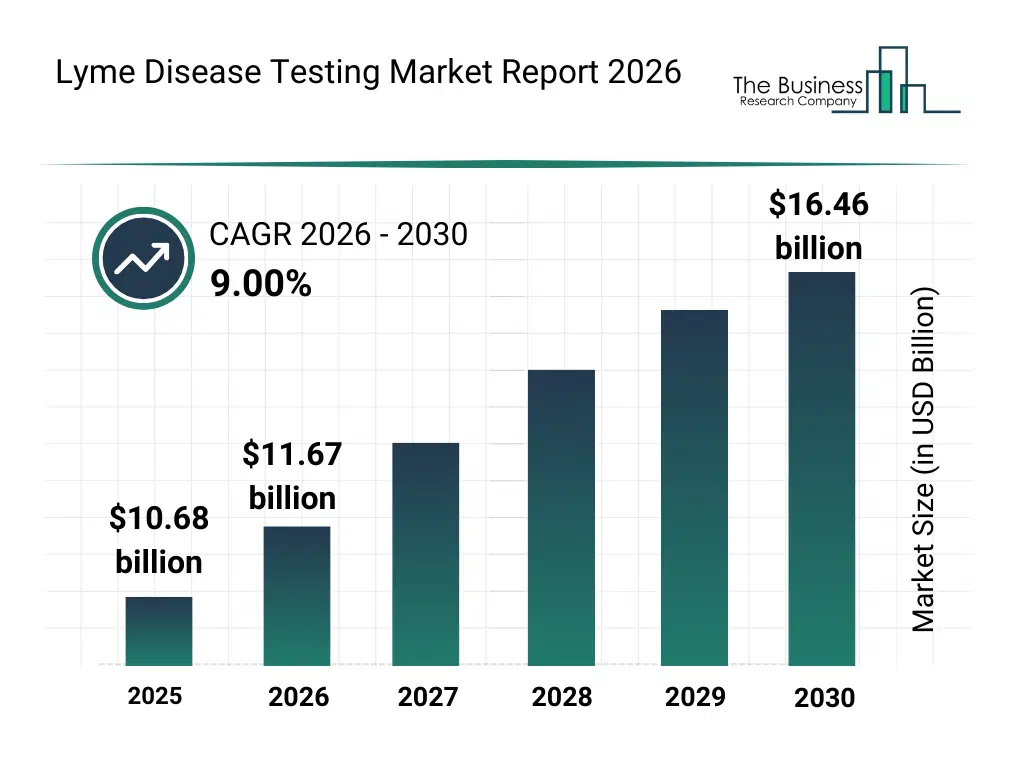
What Is The Lyme Disease Testing Market Size and Share 2026?
The lyme disease testing market size has grown strongly in recent years. It will grow from $10.68 billion in 2025 to $11.67 billion in 2026 at a compound annual growth rate (CAGR) of 9.3%. The growth in the historic period can be attributed to rising incidence of lyme disease, limited awareness about early diagnosis, reliance on conventional serological testing, increasing hospital-based diagnostics, growing research in tick-borne diseases.What Is The Lyme Disease Testing Market Growth Forecast?
The lyme disease testing market size is expected to see strong growth in the next few years. It will grow to $16.46 billion in 2030 at a compound annual growth rate (CAGR) of 9.0%. The growth in the forecast period can be attributed to adoption of ai-assisted diagnostics, growth in home-testing kits, expansion of multi-disease panels, increasing government funding for tick-borne disease surveillance, integration of cloud-based laboratory data management. Major trends in the forecast period include point-of-care testing expansion, home-based lyme disease diagnostics, multi-pathogen tick-borne panels, rapid test kits adoption, personalized diagnostic protocols.Global Lyme Disease Testing Market Segmentation
1) By Test Type: Serological Tests, Nucleic Acid Tests, Urine Antigen Tests, Other Test Types 2) By Testing Methodology: Enzyme-Linked Immunosorbent Assay (ELISA), Weste Blot, Polymerase Chain Reaction (PCR), Immunofluorescence Assay (IFA), Other Emerging Technologies 3) By Disease Stage: Early Lyme Disease, Disseminated Lyme Disease, Chronic Lyme Disease, Asymptomatic Lyme Disease Detection 4) By Sample Type: Blood, Urine, Cerebrospinal Fluid(CSF), Other Blood Samples 5) By End-User: Hospitals And Clinics, Diagnostic Laboratories, Home Care Settings, Research Institutions, Public Health Organizations Subsegments: 1) By Serological Tests: Enzyme-Linked Immunosorbent Assay, Western Blot, Immunofluorescence Assay (IFA), Line Immunoassay (LIA) 2) By Nucleic Acid Tests: Polymerase Chain Reaction (PCR), Loop-Mediated Isothermal Amplification (LAMP), Next-Generation Sequencing (NGS) 3) By Urine Antigen Tests: Lateral Flow Assay, Enzyme Immunoassay (EIA), Immunoblot Urine Tests 4) By Other Test Types: Culture-Based Tests, Tick-Borne Co-Infection Panels, Artificial Intelligence-Assisted Diagnostic ToolsWhat Is The Driver Of The Lyme Disease Testing Market?
The rising prevalence of tick-borne illnesses is expected to propel the growth of the lyme disease testing market going forward. Tick-borne illnesses refer to a group of diseases that are transmitted to humans through the bite of infected ticks, which are small, blood-feeding parasites found in grassy, wooded, or rural areas. The rising demand for tick-borne illness treatments is fueled by the expanding geographic range of ticks caused by climate change, leading to higher exposure and infection rates. Tick-borne illnesses contribute to the demand for Lyme disease testing by serving as the primary mode of transmission for Lyme disease, necessitating early detection and diagnosis to manage rising infection cases effectively. For instance, in March 2025, according to the U.S. Centers for Disease Control and Prevention (CDC), a U.S.-based federal public health agency, in 2023, over 89,000 cases of Lyme disease were reported, marking a continued high burden of tick?borne illnesses compared to previous years. Therefore, the rising prevalence of tick-borne illnesses is driving the growth of the lyme disease testing industry.Key Players In The Global Lyme Disease Testing Market
Major companies operating in the lyme disease testing market are Roche Holding AG, Thermo Fisher Scientific Inc., Labcorp Holdings Inc., Quest Diagnostics Incorporated, Eurofins Scientific, BIOMÉRIEUX, Quidel Corporation, Bio Rad Laboratories Inc., QIAGEN N.V., DiaSorin S.p.A, Meridian Bioscience, Trinity Biotech, T2 Biosystems, IGeneX, Viramed Biotech AG, Ceres Nanosciences, Galaxy Diagnostics, Lyme Diagnostics Ltd, Abbott Laboratories, Siemens Healthineers AG, Hologic Inc., Seegene Inc.Global Lyme Disease Testing Market Trends and Insights
Major companies operating in the Lyme disease testing market are focusing on technological advancements in diagnostic tools to enhance test accuracy, reduce turnaround times, and enable early-stage detection of the disease. Diagnostic tools refer to medical devices, kits, or technologies used to detect the presence of Lyme disease by identifying specific antibodies, antigens, or genetic material associated with the Borrelia burgdorferi bacteria. For instance, in August 2024, EpitogenX Ltd., a UK-based biotechnology company, announced the launch of a cutting-edge Lyme Disease Diagnostic Kit. It is an ELISA-based assay crafted using their proprietary EpitoGen platform. It features ~120 distinct Borrelia epitopes, offering high specificity and sensitivity, and is tailored for research use (RUO). It supports early, strain-specific detection and can be customized for different laboratory applications. Designed for research and diagnostic labs, it's ideal for improving detection accuracy and enabling deeper insights into Lyme disease.What Are Latest Mergers And Acquisitions In The Lyme Disease Testing Market?
In May 2024, T2 Biosystems Inc., a US-based in vitro diagnostics (IVD) company, partnered with ECO Laboratory to enhance Lyme disease diagnostics. This partnership aims to accelerate the development and clinical validation of the T2Lyme Panel, a molecular diagnostic test designed for the early and accurate detection of Lyme disease. The collaboration leverages ECO Laboratory's CLIA-certified infrastructure to expand access to advanced tick-borne disease testing across the U.S. ECO Laboratory is a US-based company, a specialized diagnostic service provider in the lyme and tick-borne disease space.Regional Outlook
North America was the largest region in the lyme disease testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Lyme Disease Testing Market?
The lyme disease testing market includes revenues earned by entities by providing services such as diagnostic test kit development, laboratory testing services, polymerase chain reaction (PCR)-based testing, serological testing, and point-of-care testing solutions. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Lyme Disease Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.67 billion |
| Revenue Forecast In 2035 | $16.46 billion |
| Growth Rate | CAGR of 9.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Testing Methodology, Disease Stage, Sample Type, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Roche Holding AG, Thermo Fisher Scientific Inc., Labcorp Holdings Inc., Quest Diagnostics Incorporated, Eurofins Scientific, BIOMÉRIEUX, Quidel Corporation, Bio Rad Laboratories Inc., QIAGEN N.V., DiaSorin S.p.A, Meridian Bioscience, Trinity Biotech, T2 Biosystems, IGeneX, Viramed Biotech AG, Ceres Nanosciences, Galaxy Diagnostics, Lyme Diagnostics Ltd, Abbott Laboratories, Siemens Healthineers AG, Hologic Inc., Seegene Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
