
Minimal Residual Disease Testing Market Report 2026
Global Outlook – By Test Type (DNA - Based Test, RNA- Based Test, Immunological Test), By Technology (Flow Cytometry, Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Other Technologies), By Application (Hematological Malignancies, Leukemia, Lymphoma, Solid Tumors, Other Application), By End User (Hospitals And Specialty Clinics, Diagnostic Laboratories, Academic And Research Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Minimal Residual Disease Testing Market Overview
• Minimal Residual Disease Testing market size has reached to $1.8 billion in 2025 • Expected to grow to $3.31 billion in 2030 at a compound annual growth rate (CAGR) of 13.1% • Growth Driver: Rising Cancer Prevalence Fueling Growth In Minimal Residual Disease Testing Market • Market Trend: Advancements In Cancer Research Testing Enhance The Minimal Residual Disease Testing Market • North America was the largest region in 2025.What Is Covered Under Minimal Residual Disease Testing Market?
Minimal residual disease testing refers to a very sensitive test to detect cancer cells in the patient's body that may remain after treatment. It is used to monitor the effectiveness of cancer treatment and inform future treatment strategies. The main test types of minimal residual disease testing are DNA - based tests, RNA- based tests, and immunological tests. DNA (deoxyribonucleic acid)-based testing (also known as genetic testing) is a type of medical examination that can spot changes to your chromosomes, genes, or proteins. The various technologies include flow cytometry, polymerase chain reaction (PCR), next-generation sequencing (NGS), and others, are used in various applications such as hematological malignancies, leukemia, lymphoma, solid tumors, and others. These are used by hospitals and specialty clinics, diagnostic laboratories, academic and research institutes, and others.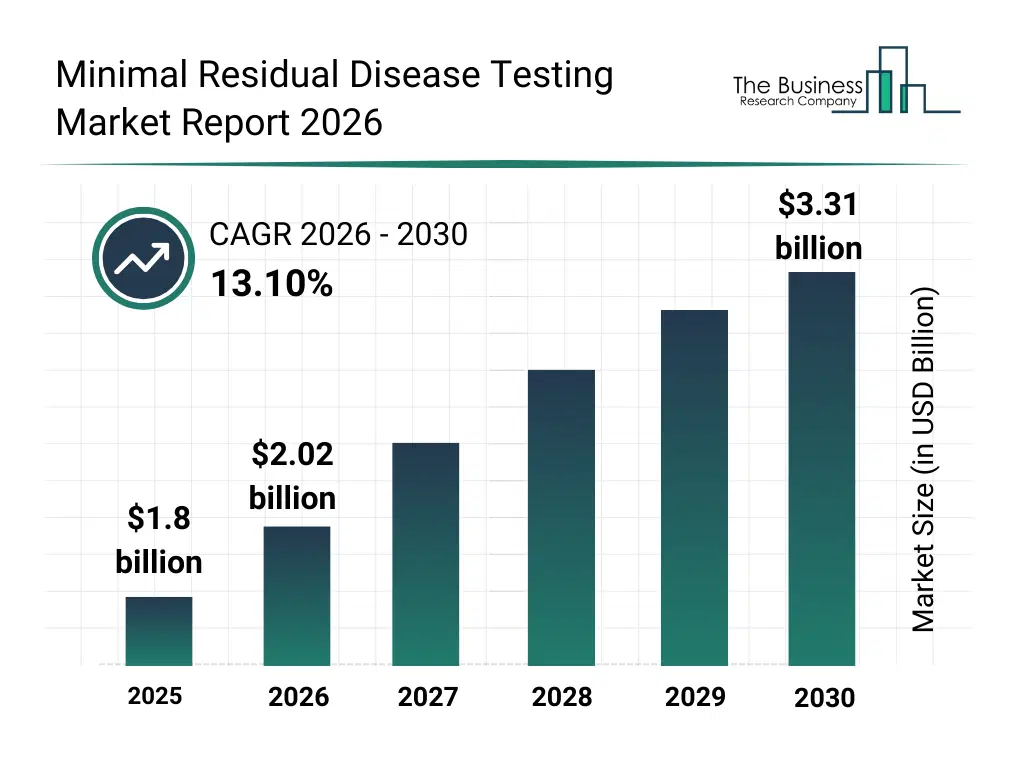
What Is The Minimal Residual Disease Testing Market Size and Share 2026?
The minimal residual disease testing market size has grown rapidly in recent years. It will grow from $1.8 billion in 2025 to $2.02 billion in 2026 at a compound annual growth rate (CAGR) of 12.5%. The growth in the historic period can be attributed to growing prevalence of hematological malignancies, advancements in molecular diagnostic technologies, rising awareness about minimal residual disease, increasing adoption of precision medicine, supportive government healthcare initiatives.What Is The Minimal Residual Disease Testing Market Growth Forecast?
The minimal residual disease testing market size is expected to see rapid growth in the next few years. It will grow to $3.31 billion in 2030 at a compound annual growth rate (CAGR) of 13.1%. The growth in the forecast period can be attributed to technological advancements in nGS and digital PCR, expansion of cancer research funding, increasing collaborations between diagnostic and pharmaceutical companies, rising demand for personalized treatment monitoring, growth in emerging markets healthcare infrastructure. Major trends in the forecast period include liquid biopsy adoption, personalized cancer monitoring, early detection & prognosis improvement, integration of molecular diagnostics, expansion of oncology research.Global Minimal Residual Disease Testing Market Segmentation
1) By Test Type: DNA - Based Test, RNA- Based Test, Immunological Test 2) By Technology: Flow Cytometry, Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Other Technologies 3) By Application: Hematological Malignancies, Leukemia, Lymphoma, Solid Tumors, Other Application 4) By End User: Hospitals And Specialty Clinics, Diagnostic Laboratories, Academic And Research Institutes, Other End Users Subsegments: 1) By DNA-Based Test: Polymerase Chain Reaction (PCR) Tests, Next-Generation Sequencing (NGS), Digital PCR 2) By RNA-Based Test: Reverse Transcription PCR (RT-PCR), RNA Sequencing, Quantitative PCR (qPCR) 3) By Immunological Test: Flow Cytometry, Enzyme-Linked Immunosorbent Assay (ELISA), ImmunohistochemistryWhat Is The Driver Of The Minimal Residual Disease Testing Market?
The increasing prevalence of cancer is expected to propel the growth of the minimal residual disease testing market going forward. Cancer refers to a condition when a few of the body's cells grow out of control and spread to other bodily regions. Cancer stages are primarily detected through minimal residual disease testing, which is a very sensitive method to detect cancer cells in the human body. It helps cancer patients prevent the disease at an early stage, boosting the minimal residual disease testing market. For instance, in January 2023, according to the National Institutes of Health (NIH), a U.S.-based government agency, in 2023, 1,958,310 new cancer cases and 609,820 cancer deaths are occured in the United States. Therefore, the increasing prevalence of cancer is driving the growth of the minimal residual disease testing industry.Key Players In The Global Minimal Residual Disease Testing Market
Major companies operating in the minimal residual disease testing market are Natera Inc., Guardant Health Inc., Laboratory Corporation of America Holdings, QIAGEN NV, F. Hoffmann-La Roche Ltd., Bio-Techne Corporation, Bio-Rad Laboratories Inc., Sysmex Corporation, Exact Sciences Corporation, GRAIL LLC, Veracyte Inc., Cergentis B.V, Mdxhealth SA, NeoGenomics Laboratories Inc., Adaptive Biotechnologies, Thermo Fisher Scientific Inc., Siemens Healthineers AG, PerkinElmer Inc., Agilent Technologies Inc., ArcherDx Inc., Asuragen Inc., Becton Dickinson and Company, Cepheid Inc., Fluidigm Corporation, Illumina Inc., Invitae Corporation, Luminex Corporation, Menarini Silicon Biosystems S.p.A.Global Minimal Residual Disease Testing Market Trends and Insights
Major companies in the minimal residual disease testing market are launching cancer research testing solutions to enhance detection sensitivity, improve treatment monitoring, and advance precision medicine approaches for better patient outcomes. The IDT Archer FUSIONPlex Core Solid Tumor Panel refers to a targeted next-generation sequencing assay used for detecting gene fusions and mutations in solid tumors, aiding in precision oncology and treatment decision-making. For instance, in April 2023, Integrated DNA Technologies (IDT), a US-based provider of genomic solutions, launched the IDT Archer FUSIONPlex Core Solid Tumor Panel, enhanced cancer research testing solution, now offering expanded and optimized coverage for single nucleotide variants (SNVs) and indels. This RNA-based sequencing assay simplifies fusion detection and variant calling in solid tumors, utilizing a single sample input and offering researchers a scalable, user-friendly solution that saves time, resources, and costs.What Are Latest Mergers And Acquisitions In The Minimal Residual Disease Testing Market?
In April 2023, Quest Diagnostics, a US-based clinical laboratory, acquired Haystack Oncology. With this acquisition Quest aims to improve personalized cancer care through Haystack's liquid biopsy technology. This approach detects residual or recurring cancer by identifying circulating tumor DNA (ctDNA) in the bloodstream. Haystack Oncology Inc is aUS-based biotechnology company.Regional Insights
North America was the largest region in the minimal residual disease testing market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Minimal Residual Disease Testing Market?
The minimal residual disease testing market includes revenues earned by entities by providing services such as peripheral blood cell testing, sequencing testing, high-throughput sequencing testing, and methylation sequencing testing. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Minimal Residual Disease Testing Market Report 2026?
The minimal residual disease testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the minimal residual disease testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Minimal Residual Disease Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.02 billion |
| Revenue Forecast In 2035 | $3.31 billion |
| Growth Rate | CAGR of 12.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Natera Inc., Guardant Health Inc., Laboratory Corporation of America Holdings, QIAGEN NV, F. Hoffmann-La Roche Ltd., Bio-Techne Corporation, Bio-Rad Laboratories Inc., Sysmex Corporation, Exact Sciences Corporation, GRAIL LLC, Veracyte Inc., Cergentis B.V, Mdxhealth SA, NeoGenomics Laboratories Inc., Adaptive Biotechnologies, Thermo Fisher Scientific Inc., Siemens Healthineers AG, PerkinElmer Inc., Agilent Technologies Inc., ArcherDx Inc., Asuragen Inc., Becton Dickinson and Company, Cepheid Inc., Fluidigm Corporation, Illumina Inc., Invitae Corporation, Luminex Corporation, Menarini Silicon Biosystems S.p.A. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
