
Bleeding Disorder Testing Market Report 2026
Global Outlook – By Product Type (Reagents And Consumables, Instruments), By Indication (Hemophilia A, Hemophilia B, Von Willebrand Disease, Idiopathic Thrombocytopenic Purpura, Other Indications), By End User (Hospitals And Clinics, Diagnostic Centers, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Bleeding Disorder Testing Market Overview
• Bleeding Disorder Testing market size has reached to $16.18 billion in 2025 • Expected to grow to $23.54 billion in 2030 at a compound annual growth rate (CAGR) of 7.8% • Growth Driver: Growth Of Bleeding Disorder Testing Market Driven By Increasing Clinical Trials • Market Trend: Innovative Gene Therapy Advancements In Bleeding Disorder Treatment Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Bleeding Disorder Testing Market?
Bleeding disorder testing refers to a range of diagnostic procedures used to evaluate the function and composition of blood to identify abnormalities related to bleeding disorders. These tests assess clotting ability, platelet function, and specific clotting factor deficiencies or dysfunctions. The main product types of bleeding disorder testing are reagents and consumables and instruments. The reagents and consumables segment of the bleeding disorder testing market refers to the various chemical substances, materials, and disposable items used in the analysis and diagnosis of bleeding disorders. These include test kits, reagents, and other consumable supplies necessary for conducting bleeding disorder tests. The various indications include hemophilia A, hemophilia B, von Willebrand disease, idiopathic thrombocytopenic purpura, and others and various treatment types involved are factor replacement therapy and drug therapy and are used by various end-users such as hospitals and clinics, diagnostic centers, and others.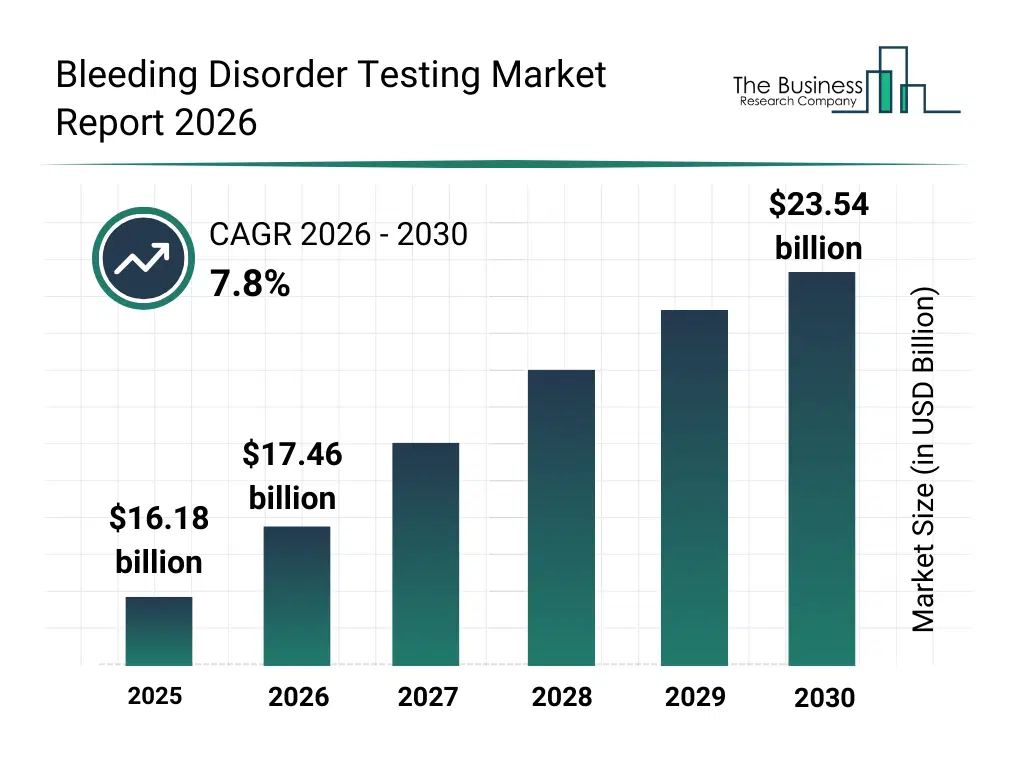
What Is The Bleeding Disorder Testing Market Size and Share 2026?
The bleeding disorder testing market size has grown strongly in recent years. It will grow from $16.18 billion in 2025 to $17.46 billion in 2026 at a compound annual growth rate (CAGR) of 7.9%. The growth in the historic period can be attributed to increasing prevalence of inherited bleeding disorders, wider adoption of laboratory diagnostics, availability of coagulation analyzers, growth of hospital diagnostic services, rising clinical awareness of bleeding disorders.What Is The Bleeding Disorder Testing Market Growth Forecast?
The bleeding disorder testing market size is expected to see strong growth in the next few years. It will grow to $23.54 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to increasing demand for rapid diagnostic solutions, expansion of personalized medicine approaches, growing investments in diagnostic automation, rising adoption of point-of-care testing, integration of ai-driven diagnostic analytics. Major trends in the forecast period include advancements in coagulation testing accuracy, growing use of point-of-care diagnostic devices, rising adoption of automated blood analyzers, expansion of genetic testing for bleeding disorders, enhanced integration of digital lab systems.Global Bleeding Disorder Testing Market Segmentation
1) By Product Type: Reagents And Consumables, Instruments 2) By Indication: Hemophilia A, Hemophilia B, Von Willebrand Disease, Idiopathic Thrombocytopenic Purpura, Other Indications 3) By End User: Hospitals And Clinics, Diagnostic Centers, Other End Users Subsegments: 1) By Reagents And Consumables: Coagulation Reagents, Plasma And Blood Collection Consumables, Hemostasis Reagents, Factor Deficiency Assay Kits, Activated Partial Thromboplastin Time (APTT) Reagents 2) By Instruments: Coagulation Analyzers, Platelet Function Analyzers, Hemostasis Testing Instruments, Point-Of-Care Testing Devices, Automated Blood Testing SystemsWhat Is The Driver Of The Bleeding Disorder Testing Market?
The increasing number of clinical trials is expected to propel the growth of the bleeding disorder testing market going forward. Clinical trials are research studies that assess the safety and efficacy of novel medical treatments, therapies, medicines, or technology in humans. The increase in clinical trials is due to advancements in medical research, growing investment in healthcare innovation, and rising demand for new treatments and therapies. Bleeding disorder testing is used in clinical trials to evaluate the efficacy and safety of new medicines and therapies for bleeding disorders, assess how well these treatments manage or correct clotting abnormalities, and monitor patient responses to ensure optimal dosing and effectiveness. For instance, according to ClinicalTrials.gov, a US-based clinical trial registry, around 477,228 clinical trials were registered at the end of 2023, an increase from 437,507 in 2022. Therefore, the increasing number of clinical trials is driving the growth of the bleeding disorder testing industry.Key Players In The Global Bleeding Disorder Testing Market
Major companies operating in the bleeding disorder testing market are Siemens Healthineers AG, F. Hoffmann-La Roche Ltd., Abbott Laboratories, Sysmex Corporation, Diagnostica Stago, Werfen Group, Sekisui Chemical Co. Ltd., Helena Laboratories, HORIBA Ltd., Mindray Medical International Limited, Nihon Kohden Corporation, Bio-Rad Laboratories Inc., Thermo Fisher Scientific Inc., Randox Laboratories Ltd., Haemonetics Corporation, QuidelOrtho Corporation, Instrumentation Laboratory, Beckman Coulter Inc., Grifols S.A., Meridian Bioscience Inc.Global Bleeding Disorder Testing Market Trends and Insights
Major companies operating in the bleeding disorder testing market are focused on developing innovative products, such as gene therapy for Hemophilia B, to provide long-term solutions, cure genetic bleeding disorders, enhance treatment outcomes, and expand their market reach. Hemophilia B is a type of bleeding disorder. It is characterized by a deficiency of clotting factor IX, which is essential for proper blood coagulation. Gene therapy for Hemophilia B is an emerging treatment aimed at providing a long-term solution by addressing the underlying genetic defect responsible for the condition. For instance, in February 2023, CSL Limited, an Australia-based biotechnology company, and the European Commission approved conditional marketing authorization (CMA) for HEMGENIX, the first and only one-time gene therapy for treating severe and moderately severe hemophilia B. This one-time treatment aims to provide long-lasting efficacy, reducing or eliminating the need for regular factor replacement therapy. HEMGENIX is notable for its innovative approach to gene therapy, offering a transformative option for patients with hemophilia B and potentially changing the standard of care in the field.What Are Latest Mergers And Acquisitions In The Bleeding Disorder Testing Market?
In February 2024, Procuritas Capital Investors VII (Procuritas), a Sweden-based private equity fund, acquired Precision BioLogic (PBI), including its subsidiary Affinity Biologicals, for an undisclosed amount. With this acquisition, Procuritas aims to expand in the hemostasis diagnostics market by leveraging Precision BioLogic’s expertise in bleeding disorder testing, including assays for hemophilia A and B, inhibitor testing, and specialty coagulation reagents. Precision BioLogic is a Canada-based clinical-stage biotechnology company specializing in diagnostic assays and quality control products specifically for bleeding disorders as part of its broader hemostasis diagnostics portfolio.Regional Outlook
North America was the largest region in the bleeding disorder testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Bleeding Disorder Testing Market?
The bleeding disorder testing market consists of revenues earned by entities by providing services such as complete blood count (CBC), prothrombin time (PT), activated partial thromboplastin time (aPTT), and genetic testing. The market value includes the value of related goods sold by the service provider or included within the service offering. The bleeding disorder testing market also includes sales of coagulation analyzers, platelet aggregation analyzers, blood collection devices, genetic testing kits, and bleeding time test kits. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Bleeding Disorder Testing Market Report 2026?
The bleeding disorder testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the bleeding disorder testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Bleeding Disorder Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $17.46 billion |
| Revenue Forecast In 2035 | $23.54 billion |
| Growth Rate | CAGR of 7.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Indication, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Siemens Healthineers AG, F. Hoffmann-La Roche Ltd., Abbott Laboratories, Sysmex Corporation, Diagnostica Stago, Werfen Group, Sekisui Chemical Co. Ltd., Helena Laboratories, HORIBA Ltd., Mindray Medical International Limited, Nihon Kohden Corporation, Bio-Rad Laboratories Inc., Thermo Fisher Scientific Inc., Randox Laboratories Ltd., Haemonetics Corporation, QuidelOrtho Corporation, Instrumentation Laboratory, Beckman Coulter Inc., Grifols S.A., Meridian Bioscience Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
