
Percutaneous Mechanical Circulatory Support Devices Market Report 2026
Global Outlook – By Product Type (Intra-Aortic Balloon Pumps, Impella Devices, Extracorporeal Membrane Oxygenation, TandemHeart, Other Types), By Application (Cardiogenic Shock, Acute Myocardial Infarction, Heart Failure, Other Applications), By End-User (Hospitals, Ambulatory Surgical Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Percutaneous Mechanical Circulatory Support Devices Market Overview
• Percutaneous Mechanical Circulatory Support Devices market size has reached to $2.46 billion in 2025 • Expected to grow to $3.87 billion in 2030 at a compound annual growth rate (CAGR) of 9.4% • Growth Driver: Rising Cardiovascular Disease Incidence Driving Growth In The Market • Market Trend: Advancements In Percutaneous Mechanical Circulatory Support Devices • North America was the largest region in 2025.What Is Covered Under Percutaneous Mechanical Circulatory Support Devices Market?
Percutaneous mechanical circulatory support (pMCS) devices are minimally invasive systems used to temporarily assist or completely replace the pumping function of the heart. These devices are inserted through a catheter into blood vessels, typically via the femoral artery, and positioned in the heart to provide mechanical support during acute heart failure, cardiogenic shock, or high-risk cardiac interventions. The main product types of percutaneous mechanical circulatory support devices are intra-aortic balloon pumps, impella devices, extracorporeal membrane oxygenation, tandemheart and other types. An intra-aortic balloon pump (IABP) is a medical device that assists the heart in pumping blood by inserting a balloon catheter into the aorta, the large artery that carries blood from the heart, to support patients with severe cardiac conditions. It is applicable to cardiogenic shock, acute myocardial infarction, heart failure, and other applications and used by various end users, including hospitals, ambulatory surgical centers, and other end users.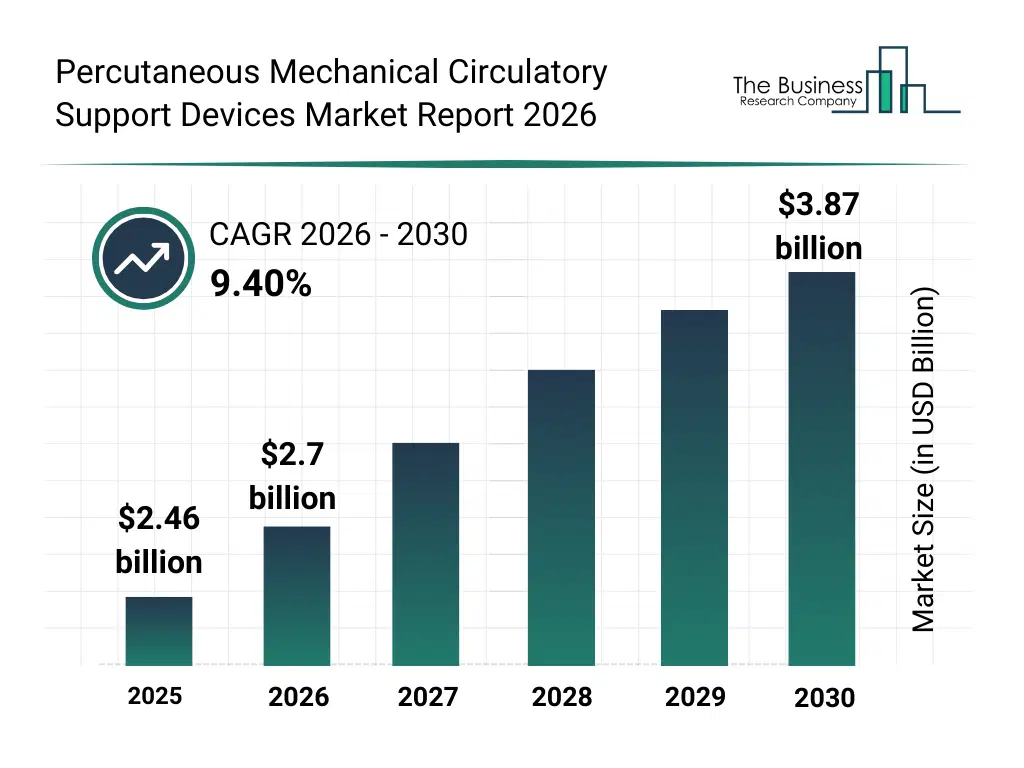
What Is The Percutaneous Mechanical Circulatory Support Devices Market Size and Share 2026?
The percutaneous mechanical circulatory support devices market size has grown strongly in recent years. It will grow from $2.46 billion in 2025 to $2.7 billion in 2026 at a compound annual growth rate (CAGR) of 9.7%. The growth in the historic period can be attributed to rising cardiovascular disease burden, increasing cardiogenic shock cases, adoption of minimally invasive procedures, ICU care expansion, aging population.What Is The Percutaneous Mechanical Circulatory Support Devices Market Growth Forecast?
The percutaneous mechanical circulatory support devices market size is expected to see strong growth in the next few years. It will grow to $3.87 billion in 2030 at a compound annual growth rate (CAGR) of 9.4%. The growth in the forecast period can be attributed to AI-assisted cardiac monitoring, growth in complex cardiac interventions, expansion of cath lab procedures, demand for temporary heart support, advanced pump technologies. Major trends in the forecast period include minimally invasive cardiac support, short-term hemodynamic assistance, high-risk pci support devices, rapid deployment cardiac systems, enhanced patient safety designs.Global Percutaneous Mechanical Circulatory Support Devices Market Segmentation
1) By Product Type: Intra-Aortic Balloon Pumps, Impella Devices, Extracorporeal Membrane Oxygenation, TandemHeart, Other Types 2) By Application: Cardiogenic Shock, Acute Myocardial Infarction, Heart Failure, Other Applications 3) By End-User: Hospitals, Ambulatory Surgical Centers, Other End Users Subsegments: 1) By Intra-Aortic Balloon Pumps (IABP): Standard IABP Devices, Enhanced IABP Devices 2) By Impella Devices: Impella 2.5, Impella 5.0, Impella CP (Cardiac Power) 3) By Extracorporeal Membrane Oxygenation (ECMO): Veno-Venous ECMO (VV ECMO), Veno-Arterial ECMO (VA ECMO) 4) By Tandemheart: Tandemheart PUMP System, Tandemheart PRO System 5) By Other Types: Centrifugal Pumps, Pneumatic Assist DevicesWhat Is The Driver Of The Percutaneous Mechanical Circulatory Support Devices Market?
The increasing incidence of cardiovascular diseases is expected to propel the growth of the percutaneous mechanical circulatory support devices market going forward. Cardiovascular diseases (CVDs) refer to a group of disorders affecting the heart and blood vessels, including conditions such as coronary artery disease, heart attacks, heart failure, stroke, and high blood pressure. The increasing incidence of cardiovascular diseases can be attributed to factors such as an aging population, unhealthy lifestyle choices, obesity, hypertension, and genetic predisposition. Percutaneous mechanical circulatory support devices are used in cardiovascular diseases to improve cardiac function, reduce heart strain, and support recovery in critical heart conditions. For instance, in October 2024, according to the Centers for Disease Control and Prevention, a US-based public health institute, in 2023, about 1 out of every 6 deaths from cardiovascular diseases was among adults younger than 65 years old, and a total of 919,032 people died from cardiovascular disease, representing 1 in every 3 deaths. Therefore, the increasing incidence of cardiovascular diseases is driving the growth of the percutaneous mechanical circulatory support (MCS) devices market.Key Players In The Global Percutaneous Mechanical Circulatory Support Devices Market
Major companies operating in the percutaneous mechanical circulatory support devices market are Abbott Laboratories, Medtronic Plc, Getinge AB, Teleflex Incorporated, Biotronik, Abiomed Inc., LivaNova Plc, Bioventrix, Berlin Heart, Eurosets, Magenta Medical Ltd, SynCardia Systems LLC, CardiacAssist Inc., Jarvik Heart Inc., Procyrion, BiVACOR Inc., CardioBridge GmbH and Company, Circulite Medical Inc., NovaPump GmbH, Supira Medical Inc., Pulsatile Technologies Inc.Global Percutaneous Mechanical Circulatory Support Devices Market Trends and Insights
Major companies operating in the percutaneous mechanical circulatory support devices market are focusing on developing technologically advanced solutions such as intra-aortic axial-flow devices to enhance patient outcomes and minimize surgical risks. An intra-aortic axial-flow device is a type of percutaneous mechanical circulatory support (MCS) device designed to assist heart function by augmenting blood flow from the left ventricle to the aorta. For instance, in February 2023, Procyrion, Inc., a US-based medical device company, announced the successful first-in-human use of its Aortix pump. This innovative device is designed to prevent acute kidney injury (AKI) in patients undergoing cardiac surgery. Delivered via a catheter in the descending thoracic aorta, the Aortix pump improves renal perfusion while decreasing the heart's workload during surgery. It achieves this by enhancing both renal and systemic perfusion while simultaneously reducing the strain on the heart, making it particularly advantageous for patients with compromised left ventricular function and renal impairment. Capable of delivering up to 5.0 liters of flow per minute, the Aortix pump is intended for temporary use during procedures such as percutaneous coronary interventions (PCI).What Are Latest Mergers And Acquisitions In The Percutaneous Mechanical Circulatory Support Devices Market?
In April 2023, Abbott Laboratories, a US-based diversified healthcare and medical device company, acquired Cardiovascular Systems, Inc. (CSI) for approximately US$890 million. With this acquisition, Abbott aims to enhance its vascular portfolio by adding CSI’s leading orbital atherectomy systems for the treatment of complex peripheral and coronary artery disease and by gaining access to an early-stage pipeline of complementary vascular intervention technologies, including catheter-based solutions for percutaneous mechanical circulatory support in high-risk coronary procedures. Cardiovascular Systems, Inc. is a US-based medical device company focused on developing and commercializing innovative atherectomy systems and related interventional technologies for the treatment of peripheral and coronary artery disease.Regional Outlook
North America was the largest region in the percutaneous mechanical circulatory support devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Percutaneous Mechanical Circulatory Support Devices Market?
The percutaneous mechanical circulatory support (MCS) devices market consists of sales of left ventricular assist devices, centrifugal flow pumps, and percutaneous right ventricular assist devices. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Percutaneous Mechanical Circulatory Support Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.7 billion |
| Revenue Forecast In 2035 | $3.87 billion |
| Growth Rate | CAGR of 9.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Medtronic Plc, Getinge AB, Teleflex Incorporated, Biotronik, Abiomed Inc., LivaNova Plc, Bioventrix, Berlin Heart, Eurosets, Magenta Medical Ltd, SynCardia Systems LLC, CardiacAssist Inc., Jarvik Heart Inc., Procyrion, BiVACOR Inc., CardioBridge GmbH and Company, Circulite Medical Inc., NovaPump GmbH, Supira Medical Inc., Pulsatile Technologies Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
