
Rituximab Biosimilars Market Report 2026
Global Outlook – By Route Of Administration (Subcutaneous, Intravenous), By Application (Non-Hodgkin's Lymphoma, Chronic Lymphocytic Leukemia, Rheumatoid Arthritis, Other Applications), By Distribution Channel (Hospital Pharmacy, Retail Pharmacy, Mail Order) – Market Size, Trends, Strategies, and Forecast to 2035
Rituximab Biosimilars Market Overview
• Rituximab Biosimilars market size has reached to $3.42 billion in 2025 • Expected to grow to $6.26 billion in 2030 at a compound annual growth rate (CAGR) of 13% • Growth Driver: Rising Non-Hodgkin's Lymphoma Cases Drive Demand For Rituximab Biosimilars • Market Trend: Collaborative Innovations In The Biosimilars Industry Are Expanding Access To Rituximab Biosimilars • North America was the largest region in 2025 and Middle East is the fastest growing region.What Is Covered Under Rituximab Biosimilars Market?
Rituximab biosimilar refers to a chimeric monoclonal antibody that is directed against the immune system's B cell-specific protein CD20. Rituximab kills B cells, making it useful for treating conditions where there are too many, too active, or otherwise abnormal B cells. The main routes of administration for rituximab biosimilars are subcutaneous, intravenous, and molecular. Intravenous therapy is a medical procedure in which fluids, drugs, and nutrients are delivered straight into a patient's vein. The different applications include non-Hodgkin’s lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, and others, and are distributed through various channels such as hospital pharmacies, online pharmacies, retail pharmacies, and other direct distribution channels.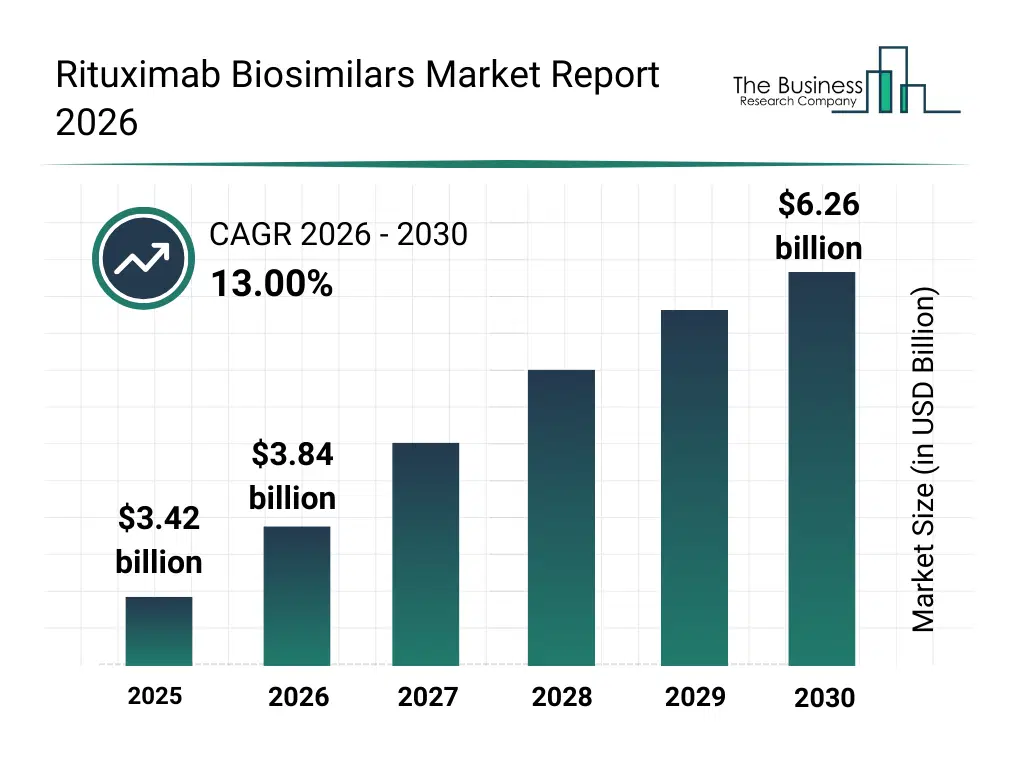
What Is The Rituximab Biosimilars Market Size and Share 2026?
The rituximab biosimilars market size has grown rapidly in recent years. It will grow from $3.42 billion in 2025 to $3.84 billion in 2026 at a compound annual growth rate (CAGR) of 12.5%. The growth in the historic period can be attributed to high cost of originator rituximab, increasing prevalence of non-hodgkin's lymphoma, growing incidence of chronic lymphocytic leukemia, rising awareness of autoimmune disease management, limited access to advanced biologics.What Is The Rituximab Biosimilars Market Growth Forecast?
The rituximab biosimilars market size is expected to see rapid growth in the next few years. It will grow to $6.26 billion in 2030 at a compound annual growth rate (CAGR) of 13.0%. The growth in the forecast period can be attributed to launch of new biosimilar formulations, expansion of subcutaneous delivery options, increasing adoption in emerging markets, rising government and insurance support for biosimilars, integration with hospital and specialty pharmacy programs. Major trends in the forecast period include rising adoption of rituximab biosimilars for oncology and autoimmune diseases, expansion of subcutaneous administration and pre-filled delivery systems, growing preference for cost-effective monoclonal antibody alternatives, increasing use of antibody-drug conjugates (adcs) in targeted therapy, enhanced hospital and retail pharmacy distribution channels.Global Rituximab Biosimilars Market Segmentation
1) By Route Of Administration: Subcutaneous, Intravenous 2) By Application: Non-Hodgkin's Lymphoma, Chronic Lymphocytic Leukemia, Rheumatoid Arthritis, Other Applications 3) By Distribution Channel: Hospital Pharmacy, Retail Pharmacy, Mail Order Subsegments: 1) By Subcutaneous: Pre-filled Syringes, Injection Pens 2) By Intravenous: IV Infusion Solutions, IV Push FormulationsWhat Are The Drivers Of The Rituximab Biosimilars Market?
The rising prevalence of non-hodgkin's lymphoma (NHL) is expected to propel the growth of the rituximab biosimilars market going forward. Non-Hodgkin's lymphoma refers to a type of cancer that starts in white blood cells, specifically lymphocytes, which are part of the body’s immune system. The rise in prevalence of non-Hodgkin's lymphoma is fueled by increasing incidence rates, particularly in aging populations and due to lifestyle and environmental factors. Rituximab Biosimilars effectively target and destroy CD20-positive B cells, helping to reduce tumor burden, improve immune system regulation, and provide a more accessible and cost-effective treatment option for patients with non-Hodgkin's lymphoma (NHL), thereby enhancing overall therapeutic outcomes and quality of life. For instance, in January 2024, according to the American Cancer Society, a US-based cancer advocacy organization, approximately 80,620 individuals (44,590 men and 36,030 women) were expected to be diagnosed with non-Hodgkin lymphoma, with an estimated 20,140 deaths (11,780 men and 8,360 women), and the lifetime risk of developing this cancer is about 1 in 42 for men and 1 in 52 for women. Therefore, the rising prevalence of non-Hodgkin's lymphoma is driving the growth of the rituximab biosimilars industry. The rise in the prevalence of autoimmune diseases is expected to propel the growth of the rituximab biosimilars market going forward. Autoimmune diseases refer to a disorder where the body's immune system assaults healthy tissues because it perceives them as foreign. Rituximab biosimilars are used to treat autoimmune diseases by targeting specific B cells, reducing inflammation and autoimmune responses. For instance, in February 2024, according to the Arthritis Australia, an Australia-based non-profit organization, an estimated 562,378 Australians will be living with RA in 2025, accounting for 14% of all arthritis cases that year. By 2040, this number is expected to rise by 33% to 748,721 people. This represents an additional 186,343 Australians living with RA in 2040 compared with 2025. Therefore, the rise in the prevalence of autoimmune diseases is driving the growth of the rituximab biosimilars industry.Key Players In The Global Rituximab Biosimilars Market
Major companies operating in the rituximab biosimilars market are Novartis AG, Pfizer, BIOCAD, Shanghai Henlius Biotech Inc., Innovent Biologics Inc., Cadila Pharmaceuticals, Hetero Drugs Limited, Dr. Reddy’s Laboratories, Shanghai Fosun Pharmaceutical (Group) Co. Ltd., Reliance Life Sciences India, Zenotech Laboratories, Celltrion, Biocad, MABION S.A., Amgen, Teva Pharmaceuticals, Celltrion Healthcare, Coherus BioSciences, Aryogen Biopharma, TR-Pharm, Hikma Pharmaceuticals plc.Global Rituximab Biosimilars Market Trends and Insights
Major companies operating in the rituximab biosimilars market are focusing on developing innovative products through strategic collaborations to enhance accessibility and provide more affordable treatment options for patients with various immunological and oncological conditions. Collaborations with established partners help companies accelerate commercialization, leverage combined expertise in regulatory approvals and distribution, and effectively bring biosimilars to market, increasing patient access and market penetration. For instance, in July 2023, Dr. Reddy's Laboratories collaborated with its partner Fresenius Kabi to commercialize its proposed biosimilar of rituximab in the US. This collaboration aims to bring a biosimilar version of rituximab to the market, providing a more affordable and accessible treatment option for patients. The availability of a biosimilar version of rituximab in the United States can potentially increase access to this important treatment option for patients with various conditions, including rheumatoid arthritis, non-Hodgkin's lymphoma, chronic lymphocytic leukemia, pemphigus vulgaris, granulomatosis with polyangiitis, and microscopic polyangiitis.Regional Insights
North America was the largest region in the rituximab biosimilars market in 2025. Middle East is expected to be the fastest-growing region in the rituximab biosimilars market report during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Rituximab Biosimilars Market?
The rituximab biosimilar market consists of sales of mabthera, truxima, ruxience, or rixathon. Values in this market are ''factory gate values,'' that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Rituximab Biosimilars Market Report 2026?
The rituximab biosimilars market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the rituximab biosimilars industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Rituximab Biosimilars Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.84 billion |
| Revenue Forecast In 2035 | $6.26 billion |
| Growth Rate | CAGR of 12.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Route Of Administration, Application, Distribution Channel |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Novartis AG, Pfizer, BIOCAD, Shanghai Henlius Biotech Inc., Innovent Biologics Inc., Cadila Pharmaceuticals, Hetero Drugs Limited, Dr. Reddy’s Laboratories, Shanghai Fosun Pharmaceutical (Group) Co. Ltd., Reliance Life Sciences India, Zenotech Laboratories, Celltrion, Biocad, MABION S.A., Amgen, Teva Pharmaceuticals, Celltrion Healthcare, Coherus BioSciences, Aryogen Biopharma, TR-Pharm, Hikma Pharmaceuticals plc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
