Biosimilar Contract Manufacturing Market Report 2026

Biosimilar Contract Manufacturing Market Report 2026
Global Outlook – By Product Type (Monoclonal Antibodies, Insulin, Erythropoietin, Granulocyte Colony-Stimulating Factor, Other Product Types), By Service Type (Process Development, Analytical And Quality Control, Fill And Finish Operations, Packaging, Other Service Types), By Source (Mammalian, Non-mammalian), By Application (Oncology, Autoimmune Diseases, Blood Disorders, Growth Hormone Deficiency, Other Applications), By End-Use (Pharmaceutical Companies, Biotech Companies) – Market Size, Trends, Strategies, and Forecast to 2035
Biosimilar Contract Manufacturing Market Overview
• Biosimilar Contract Manufacturing market size has reached to $9.94 billion in 2025 • Expected to grow to $20.35 billion in 2030 at a compound annual growth rate (CAGR) of 15.4% • Growth Driver: Growing Prevalence Of Diabetes Fueling The Growth Of The Market Due To Rising Demand For Affordable Insulin And Biologic Therapies • Market Trend: Single-Use Biomanufacturing Reduces Costs And Shortens Production Timelines • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Biosimilar Contract Manufacturing Market?
Biosimilar contract manufacturing refers to the outsourcing of biosimilar drug production to specialized third-party manufacturers with advanced infrastructure, technical expertise, and regulatory experience. These manufacturers produce biosimilars, biologic medicines highly similar to approved reference biologics in terms of safety, efficacy, and quality. It enables biopharmaceutical companies to minimize production costs, ensure compliance, and accelerate time-to-market. The main product types of biosimilar contract manufacturing are monoclonal antibodies, insulin, erythropoietin, granulocyte colony-stimulating factor, and others. Monoclonal antibodies refer to laboratory-made, highly specific proteins engineered to recognise and bind a single target (antigen) on cells or molecules, enabling precise diagnosis or therapy. They offer various services such as process development, analytical and quality control, fill and finish operations, packaging, and others with source systems, including mammalian and non-mammalian. It finds applications in oncology, autoimmune diseases, blood disorders, growth hormone deficiency, and other, serving end users such as pharmaceutical companies and biotech companies.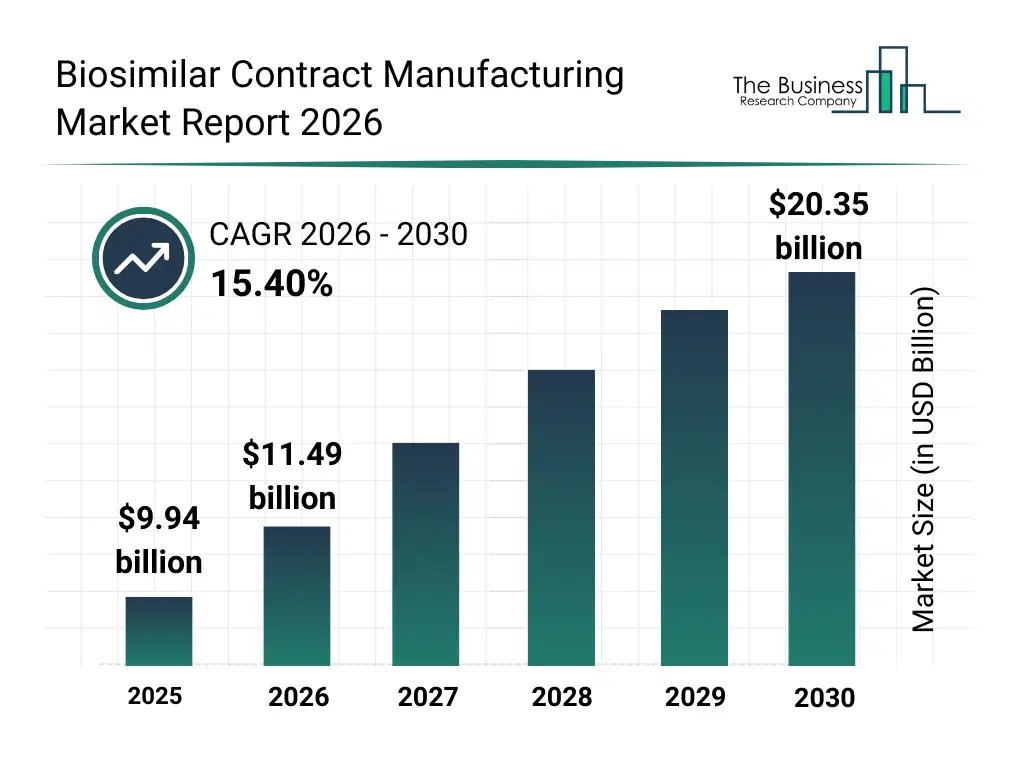
What Is The Biosimilar Contract Manufacturing Market Size and Share 2026?
The biosimilar contract manufacturing market size has grown rapidly in recent years. It will grow from $9.94 billion in 2025 to $11.49 billion in 2026 at a compound annual growth rate (CAGR) of 15.7%. The growth in the historic period can be attributed to expiration of reference biologic patents, early adoption of biosimilar development programs, expansion of contract manufacturing partnerships, increasing regulatory guidance on biosimilars, growth in chronic disease prevalence.What Is The Biosimilar Contract Manufacturing Market Growth Forecast?
The biosimilar contract manufacturing market size is expected to see rapid growth in the next few years. It will grow to $20.35 billion in 2030 at a compound annual growth rate (CAGR) of 15.4%. The growth in the forecast period can be attributed to increasing biosimilar approvals across global markets, rising pressure to reduce biologic treatment costs, expansion of biosimilar pipelines, growing investments in large-scale manufacturing capacity, advancements in process optimization technologies. Major trends in the forecast period include increasing outsourcing of biosimilar manufacturing, rising demand for cost-efficient production platforms, growing focus on analytical and quality control services, expansion of mammalian cell-based manufacturing, enhanced emphasis on regulatory compliance.Global Biosimilar Contract Manufacturing Market Segmentation
1) By Product Type: Monoclonal Antibodies, Insulin, Erythropoietin, Granulocyte Colony-Stimulating Factor, Other Product Types 2) By Service Type: Process Development, Analytical And Quality Control, Fill And Finish Operations, Packaging, Other Service Types 3) By Source: Mammalian, Non-mammalian 4) By Application: Oncology, Autoimmune Diseases, Blood Disorders, Growth Hormone Deficiency, Other Applications 5) By End-Use: Pharmaceutical Companies, Biotech Companies Subsegments: 1) By Monoclonal Antibodies: Chimeric Monoclonal Antibodies, Humanized Monoclonal Antibodies, Fully Human Monoclonal Antibodies, Bispecific Monoclonal Antibodies 2) By Insulin: Rapid Acting Insulin, Short Acting Insulin, Intermediate Acting Insulin, Long Acting Insulin, Premixed Insulin 3) By Erythropoietin: Alpha Erythropoietin, Beta Erythropoietin, Darbepoetin Alfa, Continuous Erythropoietin Receptor Activators 4) By Granulocyte Colony-Stimulating Factor: Filgrastim, Pegfilgrastim, Lenograstim, Long Acting Granulocyte Colony-Stimulating Factor 5) By Other Product Types: Interferons, Interleukins, Fusion Proteins, Enzyme Therapeutics, Hormonal BiologicsWhat Is The Driver Of The Biosimilar Contract Manufacturing Market?
The growing prevalence of diabetes is expected to propel the growth of the biosimilar contract manufacturing market going forward. Diabetes prevalence refers to the proportion of people in a population living with diabetes at a given time, reflecting ongoing treatment need and lifetime therapy demand. The rise in diabetes prevalence is due to increasing sedentary lifestyles and poor dietary habits that lead to obesity, insulin resistance, and higher rates of type 2 diabetes. Biosimilar contract manufacturing supports diabetes management by enabling the large-scale, cost-effective production of biosimilar insulin and related biologics. It ensures consistent quality and regulatory compliance, making advanced therapies more accessible to patients. This improves treatment affordability and enhances global access to essential diabetes care. For instance, in June 2024, according to the National Health Service, a UK-based government department, 3,615,330 individuals registered with a general practitioner (GP) were diagnosed with non-diabetic hyperglycemia or pre-diabetes in 2023, marking an 18% increase from 3,065,825 cases in 2022. Therefore, the growing prevalence of diabetes is driving the growth of the biosimilar contract manufacturing industry.Key Players In The Global Biosimilar Contract Manufacturing Market
Major companies operating in the biosimilar contract manufacturing market are Thermo Fisher Scientific Inc., Biocon Limited, Lonza Group AG, Samsung Biologics Co. Ltd., WuXi Biologics (Cayman) Inc., Laboratorios Farmacéuticos Rovi S.A., Vetter Pharma-Fertigung GmbH & Co. KG, National Resilience Inc., Porton Pharma Solutions Co. Ltd., Syngene International Limited, Rentschler Biopharma SE, Boehringer Ingelheim International GmbH, Enzene Biosciences Ltd., Prestige Biologics Co. Ltd., Mabion S.A., HALIX B.V., BioConnection B.V., BioCina Pty Ltd, Mycenax Biotech Inc., Polpharma Biologics S.A.Global Biosimilar Contract Manufacturing Market Trends and Insights
Major companies operating in the biosimilar contract manufacturing market are focusing on developing production facilities, such as mirrored single-use plants, to accelerate tech transfer, scale-up, and commercial reliability. Mirrored single-use plants are facilities built with identical production suites that run on disposable bioprocess equipment and unified digital systems, enabling rapid changeovers, consistent quality across sites, and lower contamination risk. For instance, in September 2025, Rezon Bio, a Poland-based contract development and manufacturing organization (CDMO), launched two mirrored-capability single-use biomanufacturing sites covering cell line development through commercial GMP supply. This approach enables faster technology transfer and flexible scale-up for biosimilars with reduced downtime, while improving batch-to-batch consistency through standardized suites, integrated analytics, and logistics automation. It also lowers the total cost of ownership for sponsors by minimizing cleaning validation requirements, shortening development and manufacturing timelines, and de-risking expansion.What Are Latest Mergers And Acquisitions In The Biosimilar Contract Manufacturing Market?
In October 2023, Advent International Corporation, a US-based private equity firm, and Warburg Pincus LLC, a US-based private equity firm, acquired BioPharma Solutions' business (now Simtra BioPharma Solutions) from Baxter International Inc. for an undisclosed amount. With this acquisition, Advent International and Warburg Pincus aim to strengthen their presence in the biopharmaceutical manufacturing sector by leveraging BioPharma Solutions’ expertise in contract development and manufacturing of biologics and biosimilars, thereby supporting global demand for advanced biopharma production capabilities. Baxter International Inc. is a US-based biotechnology company that provides biosimilar contract manufacturing services.Regional Insights
North America was the largest region in the biosimilar contract manufacturing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Biosimilar Contract Manufacturing Market?
The biosimilar contract manufacturing market consists of revenues earned by entities by providing services such as cell line development, process optimization, upstream and downstream processing, formulation, fill-finish, quality control, packaging, and regulatory support. The market value includes the value of related goods sold by the service provider or included within the service offering. The biosimilar contract manufacturing market also includes sales of biosimilar drug substances, intermediates, active pharmaceutical ingredients (APIs), finished dosage forms, cell culture media, single-use bioreactors, chromatography resins, filtration systems, and other bioprocessing consumables. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Biosimilar Contract Manufacturing Market Report 2026?
The biosimilar contract manufacturing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the biosimilar contract manufacturing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Biosimilar Contract Manufacturing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.49 billion |
| Revenue Forecast In 2035 | $20.35 billion |
| Growth Rate | CAGR of 15.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Service Type, Source, Application, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Biocon Limited, Lonza Group AG, Samsung Biologics Co. Ltd., WuXi Biologics (Cayman) Inc., Laboratorios Farmacéuticos Rovi S.A., Vetter Pharma-Fertigung GmbH & Co. KG, National Resilience Inc., Porton Pharma Solutions Co. Ltd., Syngene International Limited, Rentschler Biopharma SE, Boehringer Ingelheim International GmbH, Enzene Biosciences Ltd., Prestige Biologics Co. Ltd., Mabion S.A., HALIX B.V., BioConnection B.V., BioCina Pty Ltd, Mycenax Biotech Inc., Polpharma Biologics S.A. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Biosimilar Contract Manufacturing market was valued at $9.94 billion in 2025, increased to $11.49 billion in 2026, and is projected to reach $20.35 billion by 2030.
The global Biosimilar Contract Manufacturing market is expected to grow at a CAGR of 15.4% from 2026 to 2035 to reach $20.35 billion by 2035.
Some Key Players in the Biosimilar Contract Manufacturing market Include, Thermo Fisher Scientific Inc., Biocon Limited, Lonza Group AG, Samsung Biologics Co. Ltd., WuXi Biologics (Cayman) Inc., Laboratorios Farmacéuticos Rovi S.A., Vetter Pharma-Fertigung GmbH & Co. KG, National Resilience Inc., Porton Pharma Solutions Co. Ltd., Syngene International Limited, Rentschler Biopharma SE, Boehringer Ingelheim International GmbH, Enzene Biosciences Ltd., Prestige Biologics Co. Ltd., Mabion S.A., HALIX B.V., BioConnection B.V., BioCina Pty Ltd, Mycenax Biotech Inc., Polpharma Biologics S.A. .
Major trend in this market includes: Single-Use Biomanufacturing Reduces Costs And Shortens Production Timelines. For further insights on this market.
Request for SampleNorth America was the largest region in the biosimilar contract manufacturing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the biosimilar contract manufacturing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
