Rare Disease Clinical Trials Market Report 2026

Rare Disease Clinical Trials Market Report 2026
Global Outlook – By Phase (Phase I, Phase II, Phase III, Phase IV), By Therapeutic Area (Oncology, Cardiovascular Disorders, Neurological Disorders, Infectious Disease, Genetic Disorders, Autoimmune And Inflammation, Hematologic Disorders, Musculoskeletal Disorders, Other Therapeutic Areas), By Study Design (Interventional, Observational, Expanded Access), By End-User (Pharmaceutical Companies, Biotechnology Companies, Research Institutes, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Rare Disease Clinical Trials Market Overview
• Rare Disease Clinical Trials market size has reached to $13.3 billion in 2025 • Expected to grow to $21.34 billion in 2030 at a compound annual growth rate (CAGR) of 9.9% • Growth Driver: Surge In Personalized Medicine Fueling The Growth Of The Market Due To Improved Treatment Outcomes • Market Trend: Transformational Rebranding Highlights Commitment To Specialized Clinical Trials • North America was the largest region in 2025.What Is Covered Under Rare Disease Clinical Trials Market?
Rare disease clinical trials refer to carefully designed medical studies conducted to evaluate the safety, efficacy, and potential treatments for rare or orphan diseases that affect a small patient population. These trials aim to generate critical clinical data to support regulatory approvals and improve patient outcomes. They often face challenges such as limited patient availability, specialized protocols, and higher costs compared to trials for more common diseases. The main phases of the rare disease clinical trials include phase I, phase II, phase III, and phase IV. Phase I refers to early-stage trials conducted to evaluate the safety, dosage, and initial efficacy of investigational therapies in rare disease patients. There are therapeutic areas such as oncology, cardiovascular disorders, neurological disorders, infectious diseases, genetic disorders, autoimmune and inflammation, hematologic disorders, musculoskeletal disorders, and others. The study designs include interventional, observational, and expanded access, catering to end-users including pharmaceutical companies, biotechnology companies, research institutes, and others.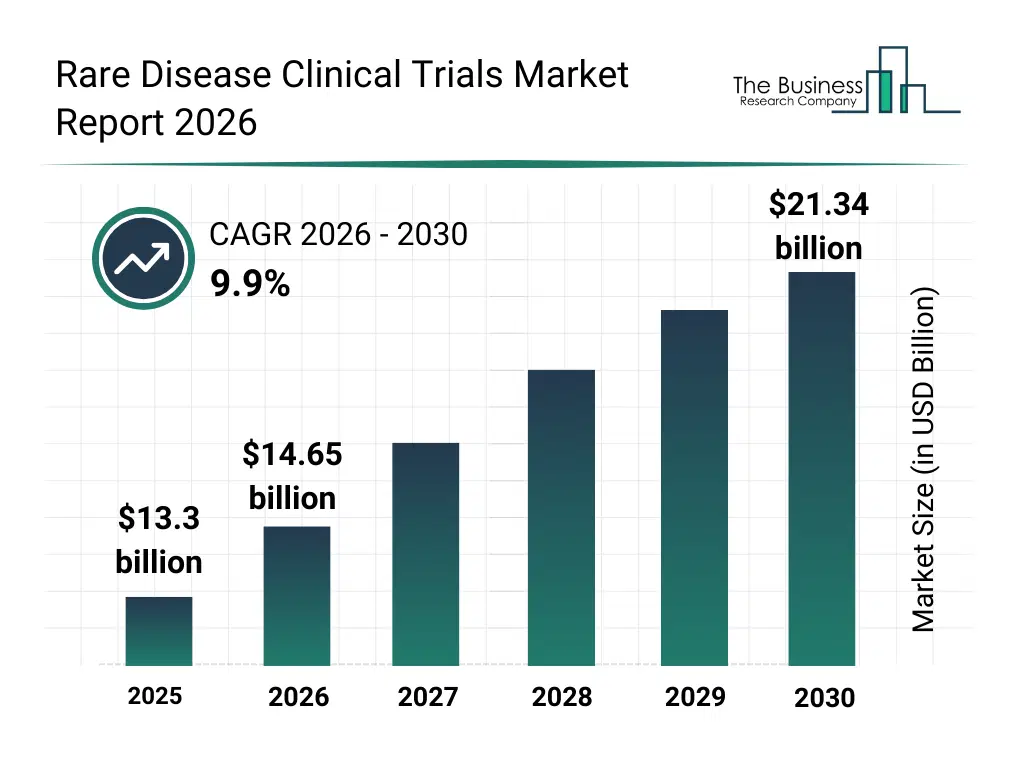
What Is The Rare Disease Clinical Trials Market Size and Share 2026?
The rare disease clinical trials market size has grown rapidly in recent years. It will grow from $13.3 billion in 2025 to $14.65 billion in 2026 at a compound annual growth rate (CAGR) of 10.1%. The growth in the historic period can be attributed to expansion of orphan drug development programs, increasing regulatory incentives for rare disease research, growth in specialized clinical research organizations, rising collaboration between academia and industry, improved diagnostic identification of rare diseases.What Is The Rare Disease Clinical Trials Market Growth Forecast?
The rare disease clinical trials market size is expected to see strong growth in the next few years. It will grow to $21.34 billion in 2030 at a compound annual growth rate (CAGR) of 9.9%. The growth in the forecast period can be attributed to increasing adoption of virtual and hybrid trials, rising use of AI-driven trial optimization, expansion of global rare disease trial networks, growing demand for faster regulatory approvals, increasing investment in rare disease R&D. Major trends in the forecast period include increasing use of decentralized clinical trial models, rising adoption of adaptive trial designs, growing integration of digital patient recruitment tools, expansion of real-world evidence collection, enhanced focus on patient-centric trial protocols.Global Rare Disease Clinical Trials Market Segmentation
1) By Phase: Phase I, Phase II, Phase III, Phase IV 2) By Therapeutic Area: Oncology, Cardiovascular Disorders, Neurological Disorders, Infectious Disease, Genetic Disorders, Autoimmune And Inflammation, Hematologic Disorders, Musculoskeletal Disorders, Other Therapeutic Areas 3) By Study Design: Interventional, Observational, Expanded Access 4) By End-User: Pharmaceutical Companies, Biotechnology Companies, Research Institutes, Other End-Users Subsegments: 1) By Phase I: First In Human Trials, Dose Escalation Studies, Safety And Tolerability Studies 2) By Phase II: Proof Of Concept Studies, Dose Response Studies, Efficacy And Safety Studies 3) By Phase III: Randomized Controlled Trials, Comparative Effectiveness Studies, Large Scale Multicenter Trials 4) By Phase IV: Post Marketing Surveillance, Long Term Safety Studies, Real World Evidence StudiesWhat Is The Driver Of The Rare Disease Clinical Trials Market?
The rise of personalized medicine is expected to propel the growth of the rare disease clinical trials market going forward. Personalized medicine refers to tailoring medical treatment to the individual characteristics of each patient, such as genetics, lifestyle, and environment. The increase in personalized medicine is due to the improved treatment outcomes, as it tailors therapies to individual patient profiles for greater efficacy and safety. Rare disease clinical trials advance personalized medicine by generating patient-specific insights that guide the development of targeted therapies tailored to each individual’s unique genetic and molecular profile. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based nonprofit organization, the Food and Drug Administration (FDA) reported that personalized medicines represented 34% of approvals in 2022, which further increased to 38% in 2023. Therefore, the rise of personalized medicine is driving the growth of the rare disease clinical trials industry.Key Players In The Global Rare Disease Clinical Trials Market
Major companies operating in the rare disease clinical trials market are F. Hoffmann-La Roche AG, Pfizer Inc., Novartis AG, AstraZeneca plc, Takeda Pharmaceutical Company Limited, IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON plc, Moderna Inc., Charles River Laboratories International Inc., Parexel International Corporation, Revvity Inc., Sarepta Therapeutics Inc., TFS HealthScience, Inventiva S.A., Tonix Pharmaceuticals Holding Corp., SpringWorks Therapeutics Inc., OrphAI Therapeutics Inc., BBCR Consulting LLC, Credevo Inc.Global Rare Disease Clinical Trials Market Trends and Insights
Major companies operating in the rare disease clinical trials market are focusing on driving innovations, such as adopting a client-centric approach to deliver personalized trial designs, enhancing patient engagement, and accelerating the development of targeted therapies for rare conditions. A client-centric approach refers to prioritizing the needs, goals, and preferences of biotech and pharmaceutical clients to provide tailored and effective clinical trial solutions. For instance, in March 2025, Evestia Clinical Limited, a US-based pharmaceutical research company, was introduced as the new brand identity of EMAS Pharma. The rebranding represents a strategic move to accelerate the company’s global expansion. The company is committed to accelerating the development of therapies for rare diseases by providing expert-driven, personalized clinical trial solutions tailored to the unique challenges of these conditions. This focus is a key part of its brand identity and service offering.What Are Latest Mergers And Acquisitions In The Rare Disease Clinical Trials Market?
In July 2024, AstraZeneca, a UK-based pharmaceutical and biotechnology company, acquired Amolyt Pharma for an undisclosed amount. Through this acquisition, AstraZeneca aims to expand its rare disease portfolio by advancing innovative therapies for rare endocrine disorders and strengthening its focus on targeted treatments for patients with unmet medical needs. Amolyt Pharma SAS is a France-based company specializing in developing therapies for rare endocrine diseases and conducting clinical trials.Regional Outlook
North America was the largest region in the rare disease clinical trials market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Rare Disease Clinical Trials Market?
The rare disease clinical trials market includes revenues earned by entities through clinical trial design and protocol development, pharmacovigilance and safety services, and laboratory and biomarker services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Rare Disease Clinical Trials Market Report 2026?
The rare disease clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the rare disease clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Rare Disease Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $14.65 billion |
| Revenue Forecast In 2035 | $21.34 billion |
| Growth Rate | CAGR of 10.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Phase, Therapeutic Area, Study Design, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Pfizer Inc., Novartis AG, AstraZeneca plc, Takeda Pharmaceutical Company Limited, IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON plc, Moderna Inc., Charles River Laboratories International Inc., Parexel International Corporation, Revvity Inc., Sarepta Therapeutics Inc., TFS HealthScience, Inventiva S.A., Tonix Pharmaceuticals Holding Corp., SpringWorks Therapeutics Inc., OrphAI Therapeutics Inc., BBCR Consulting LLC, Credevo Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Rare Disease Clinical Trials market was valued at $13.3 billion in 2025, increased to $14.65 billion in 2026, and is projected to reach $21.34 billion by 2030.
request a sample hereThe global Rare Disease Clinical Trials market is expected to grow at a CAGR of 9.9% from 2026 to 2035 to reach $21.34 billion by 2035.
request a sample hereSome Key Players in the Rare Disease Clinical Trials market Include, F. Hoffmann-La Roche AG, Pfizer Inc., Novartis AG, AstraZeneca plc, Takeda Pharmaceutical Company Limited, IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON plc, Moderna Inc., Charles River Laboratories International Inc., Parexel International Corporation, Revvity Inc., Sarepta Therapeutics Inc., TFS HealthScience, Inventiva S.A., Tonix Pharmaceuticals Holding Corp., SpringWorks Therapeutics Inc., OrphAI Therapeutics Inc., BBCR Consulting LLC, Credevo Inc. .
request a sample hereMajor trend in this market includes: Transformational Rebranding Highlights Commitment To Specialized Clinical Trials. For further insights on this market.
request a sample hereNorth America was the largest region in the rare disease clinical trials market in 2025. The regions covered in the rare disease clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here