
Oncology Based In-Vivo CRO Market Report 2026
Global Outlook – By Indication (Blood Cancer, Solid Tumors, Other Indications), By Model (Syngeneic Model, Xenograft, Patient Derived Xenograft (PDX), Other Models), By Application (Pharmaceutical Companies Or Biopharma, Biotechnology Companies, Academic And Research Institutes, CROs Or Contract Research Partners) - Market Size, Trends, And Global Forecast 2026-2035
Oncology Based In-Vivo CRO Market Overview
• Oncology Based In-Vivo CRO market size has reached to $1.5 billion in 2025 • Expected to grow to $2.54 billion in 2030 at a compound annual growth rate (CAGR) of 11.2% • Growth Driver: Surging Cancer Prevalence Fuels Growth In The Oncology-Based In-Vivo CRO Market • Market Trend: Car T-Cell Therapies In The Oncology-Based In-Vivo CRO Market • North America was the largest region and fastest growing region.What Is Covered Under Oncology Based In-Vivo CRO Market?
An oncology-based in-vivo contract research organization (CRO) refers to a company hired by another company to perform complete or partial steps of running a preclinical in-vivo study (testing or with living organisms such as animals, plants, or whole cells), such as designing, managing, monitoring, and analyzing in-vivo preclinical results for oncology drugs or devices. The main indications of oncology-based in-vivo CRO are blood cancer, solid tumors, and other indications. Blood cancers refer to cancers originating from blood-forming tissues such as immune system cells, bone marrow, and others. Leukemia, lymphoma, and multiple myeloma are some of the blood cancer types. CROs offer in-vivo testing services using blood cancer-based xenografts and other models to accelerate drug development research and help drug and device sponsors get market approval. The various models adopted by oncology-based in-vivo CRO are the syngeneic model, xenograft, patient-derived xenograft (PDX), and other models that are applied in hospitals and rehabilitation centers.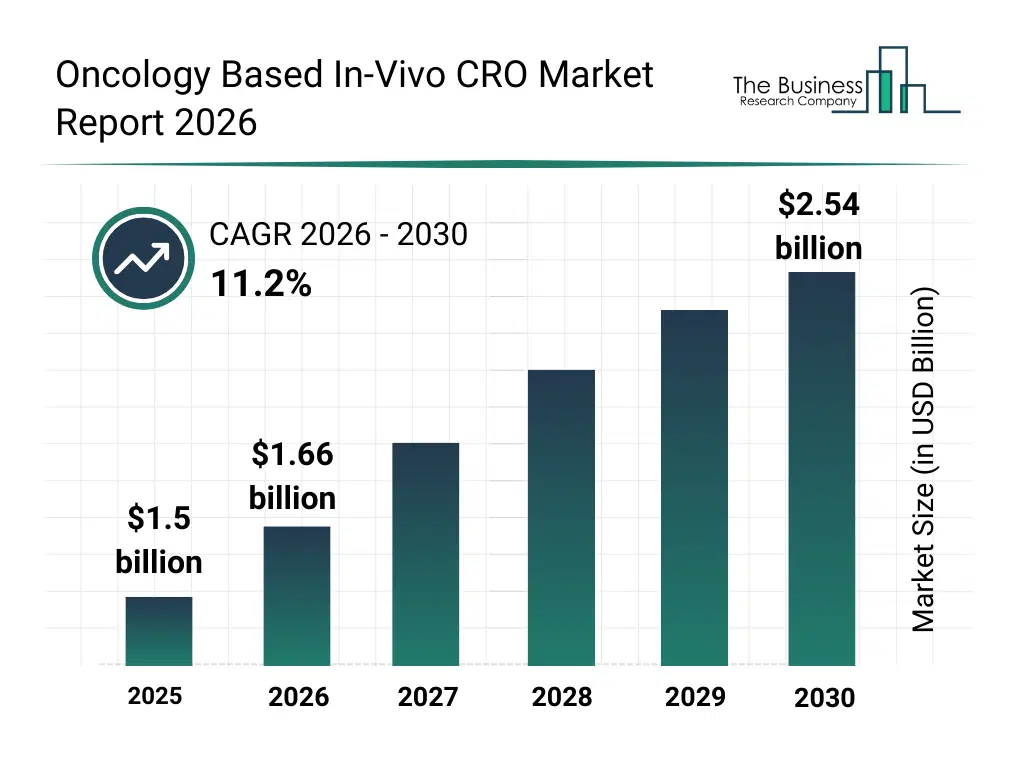
What Is The Oncology Based In-Vivo CRO Market Size and Share 2026?
The oncology based in-vivo cro market size has grown rapidly in recent years. It will grow from $1.5 billion in 2025 to $1.66 billion in 2026 at a compound annual growth rate (CAGR) of 10.6%. The growth in the historic period can be attributed to increasing demand for preclinical oncology studies, growth of animal model-based research, rising prevalence of blood cancers and solid tumors, expansion of contract research organizations globally, increasing regulatory requirements for preclinical validation.What Is The Oncology Based In-Vivo CRO Market Growth Forecast?
The oncology based in-vivo cro market size is expected to see rapid growth in the next few years. It will grow to $2.54 billion in 2030 at a compound annual growth rate (CAGR) of 11.2%. The growth in the forecast period can be attributed to rising focus on patient-derived xenograft models, increasing integration of digital data management systems, growth in personalized oncology drug development, rising outsourcing of in-vivo studies to specialized CROs, increasing adoption of automated imaging and monitoring solutions. Major trends in the forecast period include rising adoption of digital platforms for in-vivo study management, increasing use of ai and machine learning in preclinical oncology research, growing implementation of genomic profiling for patient-derived xenograft models, rising automation in in-vivo testing and data analysis, increasing iot-enabled monitoring of laboratory equipment and study outcomes.Global Oncology Based In-Vivo CRO Market Segmentation
1) By Indication: Blood Cancer, Solid Tumors, Other Indications 2) By Model: Syngeneic Model, Xenograft, Patient Derived Xenograft (PDX), Other Models 3) By Application: Pharmaceutical Companies Or Biopharma, Biotechnology Companies, Academic And Research Institutes, CROs Or Contract Research Partners Subsegments: 1) By Blood Cancer: Leukemia, Lymphoma, Myeloma 2) By Solid Tumors: Breast Cancer, Lung Cancer, Colorectal Cancer, Prostate Cancer 3) By Other Indications: Neuroblastoma, Germ Cell Tumors, SarcomasWhat Is The Driver Of The Oncology Based In-Vivo CRO Market?
The growing prevalence of cancer is expected to propel the growth of the oncology-based in-vivo CRO market going forward. Cancer refers to a group of diseases in which abnormal cells divide uncontrollably, go beyond their usual boundaries, originate in almost any organ or tissue of the body, and spread through the blood and lymph systems to other parts of the body. Treatment and diagnosis of cancer require innovative drugs and devices, which undergo different preclinical trials, clinical trials, and regulatory processes before they enter the market. An oncology in-vivo-based CRO is a company hired by a sponsor company that provides research services such as performing complete or partial steps of running a preclinical in-vivo study (testing or with living organisms such as animals, plants, or whole cells) before the drug or device is trialed in humans. With the increasing prevalence of cancer, the demand for innovative treatments and therapies has also increased, driving the growth of the oncology-based in-vivo CRO market. For instance, in April 2024, the American Cancer Society, a US-based non-profit organization, there were roughly 609,000 cancer-related deaths and 1.9 million new cancer cases identified in the country in 2022. An estimated 620,000 cancer deaths are anticipated, which would represent a little increase in mortality rates in 2024. The growing prevalence of cancer is driving the oncology-based in-vitro CRO market.Key Players In The Global Oncology Based In-Vivo CRO Market
Major companies operating in the oncology based in-vivo cro market are Crown Bioscience, Charles River Laboratories International Inc., Taconic Biosciences Inc., The Jackson Laboratory, ICON plc., WuXi AppTec Inc., Eurofins Scientific SE, Labcorp Drug Development, Champion Oncology Inc., Xentech SAS, Thermo Fisher Scientific Inc., MI Bioresearch Inc., Covance Inc., Evotec SE, Living Tumor Laboratory, Oncodesign SA, Pharmaron Beijing Co. Ltd., Syngene International Limited, Shanghai Medicilon Inc., Pharmalegacy Laboratories, Oncotest GmbH, Biocytogen LLCGlobal Oncology Based In-Vivo CRO Market Trends and Insights
Major companies operating in the oncology-based in vivo CRO market are focused on developing new solutions such as CAR T-cell therapies to gain a competitive edge in the market. CAR T-cell therapy is a form of immunotherapy that uses specially altered T cells to fight cancer. For instance, in May 2023, Seattle, a US-based Children's Hospital and Research Institute, launched BrainChild Bio to accelerate CAR T-cell therapies for children with brain tumors. The first pediatric brain tumors that BrainChild Bio would target with CAR T-cell therapy are diffuse intrinsic pontine gliomas (DIPG), an incurable childhood disease that develops in the brainstem. BrainChild Bio is a next-generation CAR T-cell treatment platform that incorporates synthetic technologies to maximize the use of CAR T-cell therapies for CNS malignancies. BrainChild Bio intends to apply its innovative CAR T-cell therapy to treat more adult and pediatric brain tumors that are challenging to cure, such as glioblastoma and brain metastases, after obtaining pediatric registration for DIPG.What Are Latest Mergers And Acquisitions In The Oncology Based In-Vivo CRO Market?
In March 2023, ClinChoice Medical Development, a US-based contract research organization (CRO) that provides research services to accelerate drug and device approvals, acquired Cromsource S.r.l. for an undisclosed amount. Through this acquisition, ClinChoice Medical Development expands its operational capabilities with additional delivery hubs to complement its existing network in Europe and the US. Data science technology offerings in several areas, including risk-based monitoring and decentralized clinical trials, are expanded through acquisition. Cromsource S.r.l. is an Italy-based contract research organization that provides full-research services to the pharmaceutical, biotechnology, and medical device industries.Regional Outlook
North America was the largest region in the oncology-based in-vivo CRO market in 2025.North America is expected to be the fastest-growing region in the global oncology-based in-vivo CRO market report during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Oncology Based In-Vivo CRO Market?
The oncology-based in-vivo CRO market includes revenues earned by entities by providing preclinical research services such as in-vivo testing via models such as Orthotopic tumor models, tumor passaging, tissue culture models, and others for oncology drugs and devices. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Oncology Based In-Vivo CRO Market Report 2026?
The oncology based in-vivo cro market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the oncology based in-vivo cro industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Oncology Based In-Vivo CRO Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.66 billion |
| Revenue Forecast In 2035 | $2.54 billion |
| Growth Rate | CAGR of 10.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Indication, Model, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Crown Bioscience, Charles River Laboratories International Inc., Taconic Biosciences Inc., The Jackson Laboratory, ICON plc., WuXi AppTec Inc., Eurofins Scientific SE, Labcorp Drug Development, Champion Oncology Inc., Xentech SAS, Thermo Fisher Scientific Inc., MI Bioresearch Inc., Covance Inc., Evotec SE, Living Tumor Laboratory, Oncodesign SA, Pharmaron Beijing Co. Ltd., Syngene International Limited, Shanghai Medicilon Inc., Pharmalegacy Laboratories, Oncotest GmbH, Biocytogen LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
