
Medical Device Clinical Trials Market Report 2026
Global Outlook – By Study Type (Feasibility And Pilot Study, Pivotal Study, FDA Premarket Approval (PMA) Application, Post-Approval Study), By Indication (Cardiovascular Devices, Neurology Devices, Orthopedic Devices, Diagnostic Imaging, Anesthesia And Respiratory Devices, Other Indications), By Study Design (Interventional, Observational, Expanded Access) – Market Size, Trends, Strategies, and Forecast to 2035
Medical Device Clinical Trials Market Overview
• Medical Device Clinical Trials market size has reached to $18.07 billion in 2025 • Expected to grow to $25.35 billion in 2030 at a compound annual growth rate (CAGR) of 7% • Growth Driver: Rising Prevalence of Chronic Diseases Fueling The Growth Of The Market Due To Increasing Demand For Innovative Diagnostic And Treatment Solutions • Market Trend: AI-Driven Innovations Enhancing Clinical Trial Design And Operational Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Device Clinical Trials Market?
Medical device clinical trials are research studies conducted to assess the safety and effectiveness of medical devices in humans. The process include phases such as feasibility, pivotal, and post-approval studies required for regulatory approval. These trials provide essential data to support device market authorization and ongoing safety monitoring. The main study types in medical device clinical trials are feasibility and pilot studies, pivotal studies, FDA premarket approval (PMA) applications, and post-approval studies. Feasibility and pilot studies are early-phase trials conducted with a small participant group to evaluate the practicality, safety, and potential challenges of a new device before advancing to larger trials. These studies are conducted across various indications such as cardiovascular, neurology, orthopedic, diagnostic imaging, anesthesia and respiratory devices, and others. Study designs typically fall under interventional, observational, and expanded access categories.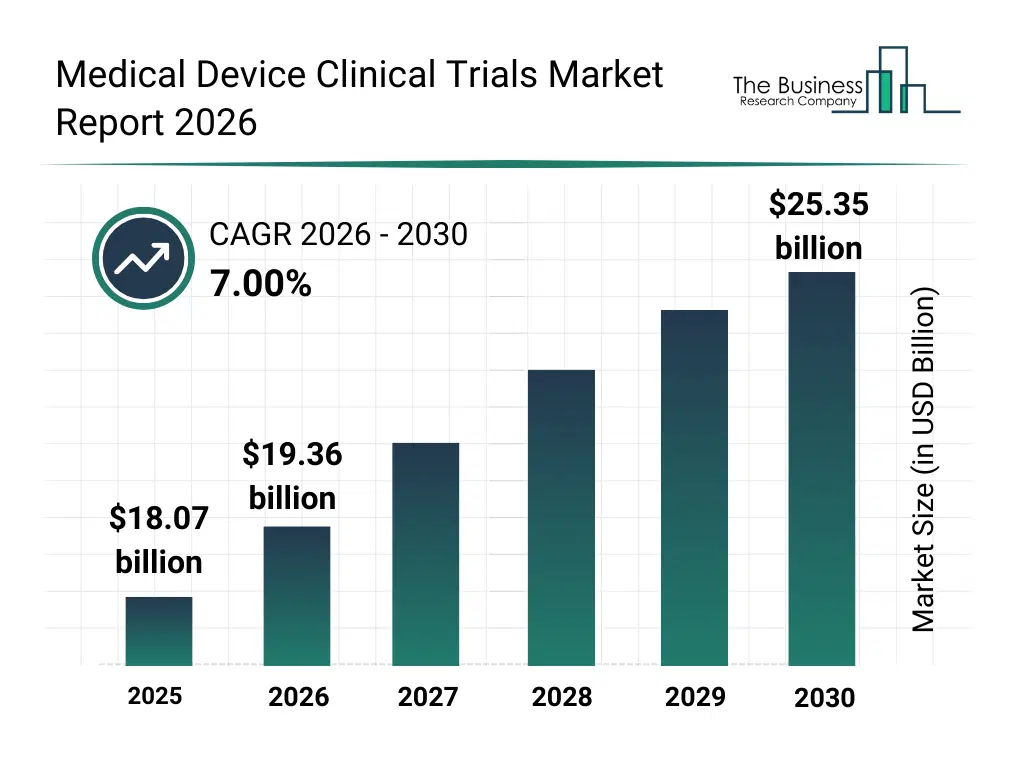
What Is The Medical Device Clinical Trials Market Size and Share 2026?
The medical device clinical trials market size has grown strongly in recent years. It will grow from $18.07 billion in 2025 to $19.36 billion in 2026 at a compound annual growth rate (CAGR) of 7.2%. The growth in the historic period can be attributed to growth in medical device innovation, stringent regulatory approval requirements, rising demand for patient safety validation, expansion of hospital-based clinical research, increasing investment in clinical evidence generation.What Is The Medical Device Clinical Trials Market Growth Forecast?
The medical device clinical trials market size is expected to see strong growth in the next few years. It will grow to $25.35 billion in 2030 at a compound annual growth rate (CAGR) of 7.0%. The growth in the forecast period can be attributed to growth in wearable and connected medical devices, increasing regulatory scrutiny on device performance, expansion of digital trial platforms, rising adoption of remote patient monitoring in trials, increasing globalization of clinical studies. Major trends in the forecast period include increasing adoption of decentralized clinical trials, growing use of real-world evidence in device trials, rising focus on post-approval and long-term safety studies, expansion of adaptive and hybrid trial designs, increasing demand for regulatory-compliant trial management.Global Medical Device Clinical Trials Market Segmentation
1) By Study Type: Feasibility And Pilot Study, Pivotal Study, FDA Premarket Approval (PMA) Application, Post-Approval Study 2) By Indication: Cardiovascular Devices, Neurology Devices, Orthopedic Devices, Diagnostic Imaging, Anesthesia And Respiratory Devices, Other Indications 3) By Study Design: Interventional, Observational, Expanded Access Subsegments: 1) By Feasibility And Pilot Study: First-in-Human (FIH) Trials, Device Safety Assessment, Procedural Feasibility Studies, Prototype Testing 2) By Pivotal Study: Comparative Effectiveness Studies, Randomized Controlled Trials (RCTs), Non-Inferiority Trials, Superiority Trials 3) By FDA PMA (Pre-Market Approval) Application: Clinical Data Submission, Device Risk Analysis, Effectiveness Evidence Studies 4) By Post-Approval Study: Long-Term Safety Monitoring, Real-World Evidence Collection, Registry Studies, Comparative Outcome StudiesWhat Is The Driver Of The Medical Device Clinical Trials Market?
The rising prevalence of chronic diseases are expected to propel the growth of the medical device clinical trials market going forward. Chronic diseases are long-term health conditions that progress slowly and require ongoing medical management. Chronic diseases are growing due to sedentary lifestyles where people spend long hours sitting at desks or using electronic devices, which reduces calorie expenditure and weakens cardiovascular health, leading to conditions such as obesity, diabetes, and hypertension. The rise in chronic diseases drives the need for medical device clinical trials to develop and validate innovative devices for effective diagnosis, treatment, and management. For instance, in June 2023, the Institute for Health Metrics and Evaluation, a US-based public health research institute, reported that over half a billion people globally were living with diabetes. This number is expected to more than double, reaching 1.3 billion people by 2050. Therefore, the rising prevalence of chronic diseases is driving the growth of the medical device clinical trials industry.Key Players In The Global Medical Device Clinical Trials Market
Major companies operating in the medical device clinical trials market are Abbott Laboratories, Siemens Healthineers, Stryker Corporation, Philips Healthcare, Baxter International, Roche Diagnostics, ICON, Intuitive Surgical, Edwards Lifesciences, Fortrea, Medidata, NAMSA, Veranex, TFS HealthScience, Avania, Parexel, Meditrial, Syneos Health, Qserve CRO, Clinius Ltd, Eclevar Medtech, 1med Sa, ISS AGGlobal Medical Device Clinical Trials Market Trends and Insights
Major companies operating in the medical device clinical trials market are focusing on developing AI-powered medical device software to optimize clinical trial protocols and resource allocation to enhance trial efficiency and reduce costs. AI-powered medical device software refers to software used in healthcare that leverages artificial intelligence (AI) algorithms especially machine learning or deep learning, to support or automate medical tasks. For instance, in January 2025, Risklick, a Switzerland-based pharmaceutical company, launched Protocol AI, an AI-based software designed to accelerate and optimize clinical trials for medical devices. Protocol AI leverages Natural Language Processing (NLP) and Machine Learning (ML) to analyze clinical data, publications, and regulatory documents, automatically drafting clinical trial protocols in minutes. This innovation significantly reduces the time and cost required for protocol development, which traditionally takes about six months, and has already demonstrated up to a 50% reduction in document development time for medicinal products.What Are Latest Mergers And Acquisitions In The Medical Device Clinical Trials Market?
In January 2024, eMed, a US-based health services company, acquired Science 37 for an amount of $38 million. This acquisition strengthens its clinical research capabilities by combining telehealth and diagnostic services with decentralized trial technology to improve enrollment and reach underrepresented patient groups. Science 37 is a US-based research company specializes in decentralized clinical trials, including medical devices.Regional Insights
North America was the largest region in the medical device clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Medical Device Clinical Trials Market?
The medical device clinical trials market includes revenues earned by entities by providing services such as clinical trial design and management, regulatory consulting, site selection and monitoring, patient recruitment, data management, and statistical analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. The medical device clinical trials market also includes sales of diagnostic devices, therapeutic devices, surgical instruments, monitoring equipment, implantable devices, wearable health technologies, and medical imaging systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Medical Device Clinical Trials Market Report 2026?
The medical device clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the medical device clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Medical Device Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $19.36 billion |
| Revenue Forecast In 2035 | $25.35 billion |
| Growth Rate | CAGR of 7.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Study Type, Indication, Study Design |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Siemens Healthineers, Stryker Corporation, Philips Healthcare, Baxter International, Roche Diagnostics, ICON, Intuitive Surgical, Edwards Lifesciences, Fortrea, Medidata, NAMSA, Veranex, TFS HealthScience, Avania, Parexel, Meditrial, Syneos Health, Qserve CRO, Clinius Ltd, Eclevar Medtech, 1med Sa, ISS AG |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
